Patient stories
Our main objective is to translate new research evidence and innovation into better patient care—not just at our Trust, but throughout London, and the world. Improving patient experience is central to why we do research.
The stories below represent first person accounts of people who have taken part in research in Northwest London and agreed to share their experience widely.
|
|
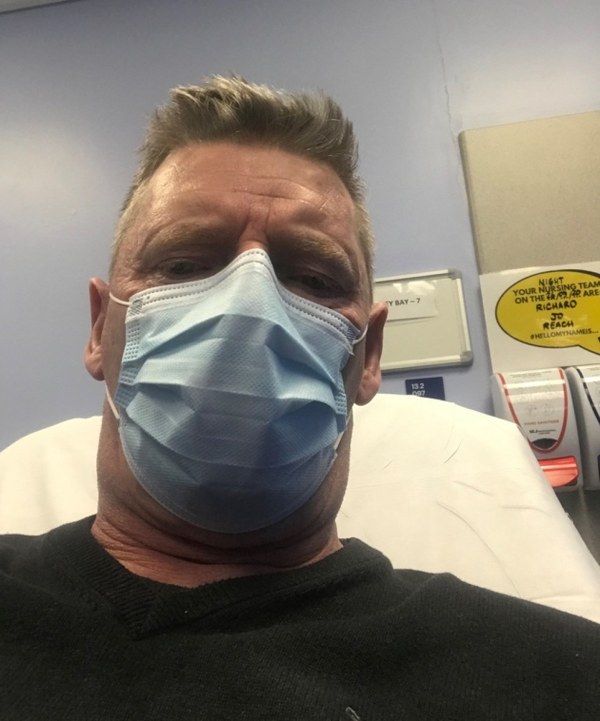
Case study: Graeme's recovery—taking part in a COVID-19 trialA man who beat coronavirus and was among the first patients to enrol on the NIHR-supported RECOVERY trial, has spoken of how he wanted to “step up” to help with research into treatments for the disease. |
|
|
Case study: Tina’s experience as a COVID-19 research volunteer at the TrustDeciding to put herself forward to take part in a COVID-19 vaccine trial was an easy decision to make for Tina, 66, from Richmond. Like a lot of volunteers, she could see that finding successful vaccines would be the key to easing the restrictions of the pandemic. |
|
|
Case study: “They make you feel very special and worthwhile”—Sharon’s experience as a COVID-19 vaccine trial volunteer at the Trust"The sooner we get vaccinated, the sooner we're all safe", Sharon recalls thinking to herself when she first decided to take part in a COVID-19 vaccine trial. The self-employed 61-year-old from London signed-up as a research volunteer at Chelsea and Westminster Hospital NHS Foundation Trust, in October last year. |
|
|
Case study: Clea gets accurate diagnosis after taking part in researchSomeone is told they have diabetes every two minutes in the UK. Clea St Jean, a Bed Manager at Imperial College Healthcare NHS Trust, knows exactly how shocking this diagnosis can be. Clea was diagnosed with type 1 diabetes. But researchers in North West London discovered she actually has a rare form of the condition. |
|
|
Case study: Frank who cared for his mother who lived with dementia is passionate about research after getting involvedFor Frank Arrojo, taking part in dementia research helped him better understand the condition. Learning more about dementia has been bittersweet for Frank. He now better understands how carers can help but wishes he had this knowledge sooner. "I always think about what if only then, when I was caring for my mum, I knew what I know now", says Frank. |
What do our patients think about participating in research?
Mr Quail, Clinical trial investigating medicine that will allow patients to recover uneventfully after surgery
“In the beginning I was ignorant to studies going on. Being a part of the research study has brought to my attention that the public are not as responsive to research as they should be. At first all of the information can be quite difficult to understand. There is a lot to take on board, but the research nurse explains everything so well. Fiona (research nurse) was lovely and really put my mind at rest. I was treated so well and I found the whole process very efficient."
“The reason I took part was because I really wanted to help. It brings me great joy to know that in the future I could be helping my relatives especially my son. Unless there are volunteers then there will be no improvements, this is the only way we can progress. I am not sure if the research has had any direct benefits for me but I hope my contribution will help somebody else in the future.”
Eric, Paediatric clinical trial
“I thought that it might not affect me much but it might help somebody else if I do it. It was an easy decision to make and I didn’t have to think too hard about it. I thought I might as well do it because all it took was an extra vile of blood. I don’t know if it will help me but I am hoping it will help someone else. I would tell somebody my age to take part because it’s not a lot of work, they care about you more and it might help someone else. You may as well do it because it’s not going to do anything bad to you.”
Eric's mother
“We have only recently received the diagnosis for Eric and there was so much to take on board. I felt like if I had known more about the whole process it would have been better for me. So if the next family that gets diagnosed with this has a little bit more information due to us taking part in research, then they can benefit from it. Selfishly I thought that Eric would be followed very closely, be monitored and receive extra attention. So for selfish reasons I wanted him to take part.
The research nurse Lauren has been incredibly helpful and calls to check on Eric, they send emails to keep in touch and meet us to make sure we know where we are going. I feel like Lauren has done a lot of hand holding which has been incredibly helpful as everything is new, she has been a personal support. We have only found benefits from taking part in the research study.”
Mrs Guerrero, Clinical trial investigating medicine that will allow patients to recover uneventfully after surgery
“I love my hospital—I have had a very nice experience here. The research staff have given me extra attention and made my experience better as I was very worried about my operation. I feel very secure with the research nurses and the staff here are first class. When I was on the ward I had the research nurses coming by to see me and my neighbours were jealous of the extra attention I was getting!"
“I wanted to take part in research because they might find something that will help other people. Science is growing all the time it’s incredible how fast everything is changing. I was so excited and emotional that little me could help with something very big, I feel very special. I think that human beings are like plants, if they do not grow you have to know why – so you have to understand how it all works to help them grow."
“I think it is important that people take part in research because it can help the people with the same illness; if people did not take part we would not know how to make things better. I would be happy to take part in research again.”
Research Experience Survey
Every year, we encourage research participants at the Trust to provide feedback on their experience of research by completing the national Participant in Research Experience Survey (PRES) devised by the National Institute of Health and Care Research.
The most recent PRES (2018/2019) highlighted that 90% of respondents had a good or better experience of taking part in clinical research!
To find out alternative ways of how to leave your feedback with our research department, email chelwest.research@nhs.net.